HOME › Research and Resources › Animal Models for Human Diseases › More Information

More Information
1. Key members
- Project leader - Taisei Nomura
- Subleader - Haruko Ryo
- Researcher - Shigeki Adachi,Hiroyuki Iwaki
2. Background and objectives
This is a project to research the development of animal model for human diseases, which play essential roles in the drug development process, jointly with pharmaceutical companies. We carry out analyses of pathologic conditions and genes, validation and verification studies for drug development, and safety studies using mice that spontaneously develop diseases, highly sensitive strains of mice that are used for safety evaluations, and super SCID mice (including long-term maintenance of mice with transplanted human organs or tissues). We intend to improve and develop easy-to-handle and reliable strains, and also to provide researchers with education and guidance with the objective of contributing to drug development.
3. Overview of our research
- Research on the pathologic conditions and genetics of mice that spontaneously develop diseases
- Development and applied research of highly sensitive mouse strains for safety evaluations
- Development of technology to directly evaluate the effects of drugs on human body using human organs and tissues
- Improvement and development of easy-to-use and reliable strains
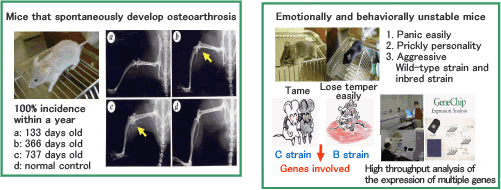
I.Fundamental research on mouse models that spontaneously develop diseases
These mouse strains allow research on (i) multiple diseases, including cancers, developmental malformations, and other fatal disorders, in an individual mouse; (ii) DNA damage and repair; and (iii) efficient detection and analysis of mutations (the number of mice required to identify a causative mutation is much smaller than that needed for conventional methods).
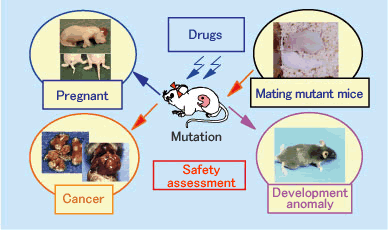
II.Development and application of highly sensitive mouse strains for the evaluation of the safety of pharmaceutical products
The physicians (or medical institutions) that provide us with clinical specimens are charged with the necessary and important function of diagnosis. We are constructing a system so that when the specimens are to be used in research, it can decide upon the suitability of both the research and oversee that the rights of the physicians of the medical institutions providing the specimens are appropriately upheld, thus ensuring these valuable materials are used reasonably and impartially in research.
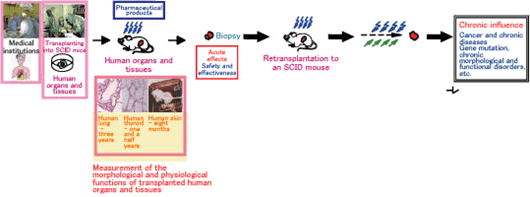
III.Development of technology to directly evaluate the influence of pharmaceutical products on the human body using human organs
TWe develop mice that can be transplanted with human organs or tissues (super SCID mice: mice that do not mount an immunological response to transplanted tissues). These mice allow researchers to perform experiments in a system that mimics the conditions in humans and to use these mice as models for drug development and safety research, genetic and immune therapies, and human cancer development and metastasis.
To inquire about the Animal Models for Human Diseases, please contact



